Differentiation between Viral and Urinary Tract Infections Using the Modified Rochester Criteria In Febrile Infants Younger than three Months
Article information
Abstract
Purpose:
The symptoms and signs of urinary tract infection (UTI) in early infancy are non-specific. Prompt diagnosis of UTI is important, as untreated UTI results in renal damage. Especially, febrile UTI in young infants coexist with other serious bacterial infections. The purpose this study was to propose modified Rochester criteria to differentiate viral infection from urinary tract infection.
Methods:
We carried out a retrospective investigation of 168 infants less than three months old with a tympanic temperature >38ºC who were admitted to Chung-Ang University Hospital between 2011 and 2014. We compared the symptoms, physical examination results, and laboratory data between viral infection and UTI groups. A modified Rochester criterion was composed of statistically significant factors.
Results:
A total of 76 and 92 infants with UTI and a viral infection, respectively, were included. Statistically significant differences in gender, previous admission history, neutrophil ratio, and urine WBC count were found between the two study groups. Using a cut off value of 3 points, the sensitivity and specificity of the modified Rochester criteria were 71.28% and 78.57%, respectively.
Conclusion:
The modified Rochester criteria may give an outline for identifying young infants with UTI.
Introduction
Urinary tract infection (UTI) is one of the most common bacterial infections in children. In infants younger than 3 months, symptoms such as fever, vomiting, and feeding refusal are nonspecific and may result in delayed diagnosis and major complications, such as renal scars. Therefore, accurate diagnosis and early appropriate treatment in this age group are important [1,2]. In general, urine analysis using urine bags is a useful and easy screening test but the false-positive rates are unacceptably high. With this method, the presence of infection is difficult to determine when microscopic examination reports are equivocal with respect to the number of white blood cells [3]. Therefore, a variety of criteria and tests have been proposed to predict urinary tract infections and complications [4].
Moreover, concomitant bacteremia is associated with UTI in young infants, and many studies have focused on the early identification of predictors of severe bacterial infection in this age group [5-7]. Before 1985, hospitalization was recommended in febrile infants less than 60 days old, for sepsis evaluation and administration of intravenous antibiotic treatment. However, during this period, the various criteria used to discriminate high-risk groups had insufficient sensitivity [8]. In addition, these recommendations had disadvantages in terms of unnecessary hospitalization, nosocomial infections, and side effects of injudicious use of antibiotics [9]. During the late 1980s and the early 1990s, new criteria were developed to distinguish low- from high-risk infant and recommended close observation without antibiotic treatment for the management of UTI in this age group. Various low-risk criteria were proposed and their utility has been validated to the present day [10]. Rochester criteria was applied for selecting infants who underwent careful observation as outpatients without receiving antibiotic therapy, with a high sensitivity, especially in young infants [10]. The Rochester criteria definition of serious bacterial infection (SBI), which typically include meningitis, bacteremia, and UTI, vary across the published literature [11].
Although the source of infection is clear in most patients visiting the hospital because of fever, the source of infection is not apparent after medical history and physical examination in 20% of febrile children. In children with fever without an identifiable source, UTI is the most common bacterial infection and occurs in 7.5% and 4% of infants younger than 8 weeks and 2 years, respectively [12]. Based on the original Rochester criteria, the purpose of this study was to propose a set of modified Rochester criteria to predict UTIs in low-risk groups, and to differentiate between viral infection and UTIs in febrile infants.
Materials and Methods
1. Patient identification
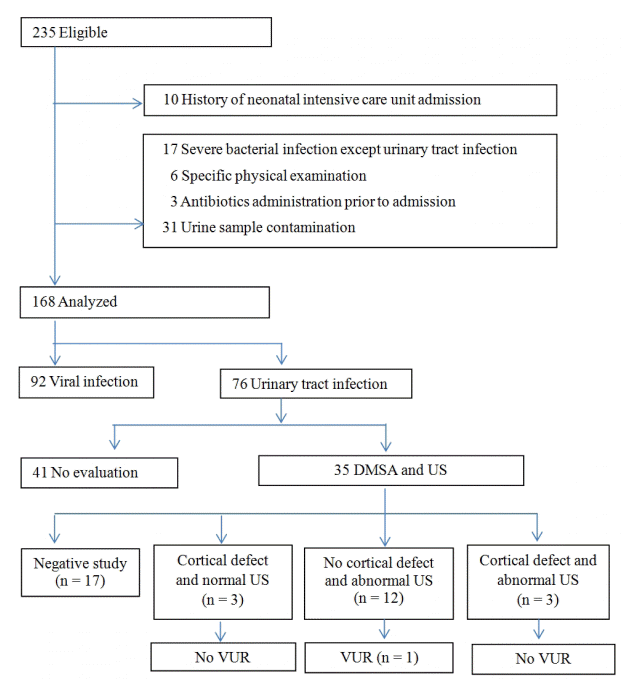
This was a retrospective review study of 235 infants less than 3 months with temperatures over 38.0ºC admitted to Chung-Ang University Hospital, between January 2011 and June 2014. We excluded 10 patients with congenital malformations and with a history of admission to the neonatal intensive care unit because of perinatal complications. Patients were also excluded if they had sepsis or meningitis and had specific physical findings suggestive of a local infection (n = 23), received antibiotics before admission (n = 3), and had a contaminated urine sample (n = 31). Finally, we analyzed data from 168 patients (Fig. 1).
2. Methods
We collected data on patient demographics, past medical history, clinical manifestations, physical examination, and laboratory findings. We defined urinary tract infections if urine cultures grew a single pathogen and had colony counts of more than 104 colony forming units (CFU)/mL in samples obtained from a catheterized specimen. Equivocal urinalysis results were defined as the number of white blood cells seen, 5–9 or 10–19, in a high power field (HPF) during urine microscopic examination.
All patients were hospitalized and treated with parenteral antibiotic therapy after a full sepsis evaluation [13]. Febrile UTI was evaluated at the earliest convenient time using renal ultrasound with 99m-Technetium dimercaptosuccinic acid (99mTc-DMSA) during hospitalization. When there were cortical defects in 99mTc-DMSA, a voiding cystourethrogram (VCUG) was performed and the results interpreted by imaging specialists.
Viral infection defined as a negative culture but a positive respiratory virus polymerase chain reaction. Some study results indicated a significantly lower prevalence of UTI in infants with viral infection or bronchiolitis versus infants free of viral infection or bronchiolitis [14,15].
Statistical analyses were carried out using SPSS 18 software (SPSS Inc, Chicago, IL, USA). The Chi-square test and Mann-Whitney U-test was used for independent variables and the Student’s t-test was used for dependent variables. The level of statistical significance was assumed to be P<0.05. A receiver operating characteristic (ROC) curve was used to evaluate the modified Rochester criteria performance.
3. Modified Rochester criteria
The components of the low-risk criteria in the original Rochester criteria include a previously healthy term infant with no history of previous antibiotic treatment, normal physical examination findings, WBC count 5,000–15,000 cells per mm3, band count <1,500 cells per mm3, and catheterized urinalysis WBC count <10 per HPF 10).
In this study, the 9 following laboratory tests were performed: C-reactive protein, peripheral WBC, neutrophil percentage, platelet counts, hemoglobin, total bilirubin and direct bilirubin ratio, aspartate aminotransferase, alanine aminotransferase, and urine microscopic exam. We also analyzed age, gender, medical history [16]. If urine microscopic examination revealed white blood cells < 1, 1–4, 5–9, 10–19, 20–29, 30–49, 50–99, more than 100 per HPF, we defined as grade 0, 1, 2, 3, 4, 5, 6, and 7 respectively. Modified Rochester criteria were composed of the statistically significant factors and the cut-off point of the ROC was used.
Results
1. Patient characteristics
A total of 76 and 92 infants were diagnosed with UTI and viral infections, respectively. The number of male infants was significantly higher in UTI than in the viral infection group (P <0.05). Statistically significant differences in age of onset were found between the two groups (P = 0.01).
The percentage of neonates in the viral and UTI group was 26.09% and 10.53%, respectively (P = 0.01). The rate of previous admission history was 7.60% and 18.42% in the viral and UTI group, respectively, and this difference was statistically significant (P = 0.035). No differences were observed between the two groups in terms of gestational age, birth weight, and type of delivery (Table 1).
2. Laboratory findings
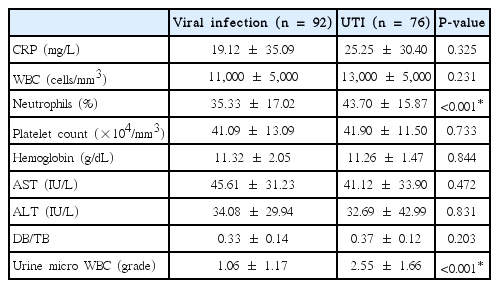
The viral infection group had a significantly higher percentage of neutrophils in peripheral blood than the UTI group (P<0.001). WBC grade on urine microscopic examination was higher in the UTI group (grade 2.55) than in the viral infection group (grade 1.06), and this difference was statistically significant (P <0.001) (Table 2). No statistically significant differences were observed in the levels of C-reactive protein, total WBC count, platelets, hemoglobin, total bilirubin and direct bilirubin ratio, aspartate aminotransferase, alanine aminotransferase, between the two groups. WBC on urine microscopic examination was <10 (/HPF) in 37 infants (48.7%) in the UTI group, and 10–19(/HPF) in 5 infants (6.57%), in the viral infection group.
3. Modified Rochester criteria for UTI
The components of the low-risk criteria in the Modified Rochester criteria were female, no previous admission history, percentage of neutrophils in white blood cells ≤40%, catheterized urinalysis <10 WBC per HPF, with each component being worth 1 point (Table 3).
When we used more than 3 points as the cut-off point, the area under the ROC curve (AUC) was 0.787 and the Modified Rochester criteria showed a sensitivity and specificity of 71.28% and 78.57%, respectively. The AUC of the original Rochester criteria was 0.733, and the sensitivity and specificity was 63.16% and 78.57%, respectively (Table 4). The AUC of the Modified Rochester criteria in 9 and 5 UTI infants with WBC counts of 5–9 and WBC 10–19 per HPF on microscopic urinalysis was 0.75 and 0.46, respectively. No statistically significant differences were observed between UTI and acute pyelonephritis (P=0.110).
Discussion
Anbar et al. reported that severe bacterial infections affected 10% of febrile infants younger than 3 months [17]. Various low-risk criteria for bacterial infection have been proposed with the Rochester criteria being one of them. McCarthy et al. indicated that from a total of 86 infants younger than 60 days, in a prospective study and to identify low risk infants by using Rochester criteria is not without risk, only one had a SBI [18], and the prospective study of Jaskiewicz et al. reported high negative predictive values, using the Rochester criteria [19].
Because the urinary tract is a common site of bacterial infections and UTI is often concurrent with bacteremia, the aim of this study was to determine whether the Rochester criteria can be used to predict the risk of UTI in young infants [20]. We performed a retrospective chart review of infants younger than 100 days and analyzed the data in 169 of those. Differences in gender, previous admission history, neutrophil percentage, and WBC on urine microscopy were statistically significant and comprised the Modified Rochester criteria.
The presence of nitrites and leukocyte esterase serves as indirect evidence of a UTI. However, the time necessary for certain bacteria to reduce nitrates to nitrites may be insufficient in infants with difficulties in toilet training, and this test may produce false-negative results.
The definitive diagnosis of an UTI requires the isolation of at least an uropathogen from urine cultures and because urine cultures typically require at least 24 hours of incubation, urinalysis and urine microscopy are often used to guide initial empiric therapy. The false-positive predictive value of pyuria (more than 5 WBC/HPF) is high and negative urine microscopy does not rule out UTI. In this study, our intention was to ascertain whether the Modified Rochester criteria could help to determine the presence of UTI in cases in which urinalysis was equivocal.
The total number of points in the Modified Rochester criteria is 4, and it appears that if more than 3 points were used as the cut-off point, the distinction between UTI from viral infection may be made. In addition, when urinalysis is equivocal, it may be useful. Because there are more urinary tract anomalies and vesicoureteral reflux in boys, and boys with foreskin harbor higher concentrations of uropathogenic microbes, they have a higher incidence of UTI during the first year of life. In this study, a 1.98-fold increased risk for developing UTI was reported [21]. During the first 3 months, UTI is often concurrent with bacteremia in approximately 10% of patients, while bacteremia with UTI was present in 1 patient in this study (1.32%).
Infants younger than 3 months are often managed by using established low-risk criteria, such as the Rochester criteria, Philadelphia criteria, or Boston criteria. All these criteria include infants younger than 3 months who show a nontoxic clinical appearance. Unlike the other two criteria, the Rochester criteria do not include lumbar puncture and chest radiographs [22]. Moreover, the Philadelphia criteria include the band/neutrophil ratio, whereas the Rochester criteria use absolute band count. Philip and Hewitt reported that the neutrophil index is a sensitive and specific predictor of bacterial infection [23].
Recently, jaundice is thought to be one of the clinical signs of UTI in the neonatal period. Although the association between neonatal jaundice and UTI is still unidentified, bilirubin levels may rise as a result of a UTI [24]. Testing for UTI has been recommended as part of the evaluation of asymptomatic jaundice. In this study, we found no association between UTI and bilirubin levels, and this is may be because young infants showed a higher incidence of UTI than neonates.
Berger et al. reported that C-reactive protein was the best test for early diagnosis of bacterial infections in newborns and for determining the period of antibiotic use. Moreover, Jung et al. showed that WBC count and C-reactive protein could predict renal defects but did not show significance in this study [25]. C-reactive protein does not have a direct relationship with kidney disease but is a very sensitive indicator of bacterial infection, and increases in patients with acute pyelonephritis, as reported in several studies [26].
In this study, 76 young infants were diagnosed with UTI, and 41 did not undergo renal ultrasonography and 99mTc-DMSA. Because urinary tract malformations are often diagnosed in the prenatal period, due to the common use of prenatal ultrasonography, most renal scars after UTI are not serious. However, whether or not imaging studies must be performed in infants with initial febrile UTI with signs and symptoms that respond well to antimicrobial therapy, remains unclear. However, results from recent studies have shown that about 4% of UTI infants younger than 2 months were diagnosed as having anatomical abnormalities on renal ultrasound [20]. Although urine culture is negative in UTI due to fluid, frequent urination, and antibiotic treatment, a cortical defect in 99mTc-DMSA may still be found. The limitation of this study is that not every infants with UTI underwent imaging studies.
In addition, acute pyelonephritis occurred in only 6 of 168 infants with UTI. We could not apply the Modified Rochester criteria to predict complications. In addition, other limitations that we were not able to address included the follow-up of infants who showed a renal cortical defect in 99mTc-DMSA, to ensure that a permanent renal scar was not developed.
If Modified Rochester criteria can predict complications of UTI such as urosepsis and cortical defect, it can bring real added value. However, during the study period, complications were rare. So, our study only made a comparison between viral infection and UTI.
In conclusion, we believe this study may be helpful for the differential diagnosis of viral infections from UTI in febrile young infants despite limitation. Because there are few studies restricted to infants younger than 100 days, and the low risk criteria varies between studies, it is difficult to select the recommendations [5,27].