A Case of Orthostatic Proteinuria Progressed to Persistent Proteinuria Associated with Renal Pathology
Article information
Abstract
Orthostatic or postural proteinuria is the most common cause of asymptomatic proteinuria in children. As orthostatic proteinuria (OP) is a benign disease with relatively good prognosis, it has no specific management, and patients only need to be observed. However, if OP shows a persistently high level of proteinuria, in theory, glomerular changes can occur. An 11-year-old girl was referred to the hospital due to asymptomatic proteinuria and was diagnosed as having OP based on the results of clinical and laboratory examinations, urinalysis, and protein/creatinine (TP/Cr) ratio at both supine and erect positions. During follow-up observation, the 24-hour TP/Cr ratio was persistently higher than 1.5 mg/mg for 2 years. We performed renal biopsy, which showed mesangial proliferative glomerular lesions with focal effacement of the podocyte foot processes, but without immune depositions. OP can be accompanied by glomerular lesions if moderate to severe proteinuria persists.
Introduction
Orthostatic proteinuria (OP) is a non-pathological form of isolated asymptomatic proteinuria, which is characterized by the presence of protein in urine samples collected in the upright/ambulatory position and its absence in urine samples collected in the supine position [1,2]. It affects 2-5% of children or young adults with asymptomatic proteinuria, but a recent report suggested a very high incidence (≥15%) in children between 6 and 18 years [3-5]. In a Korean report on school health screening, OP is the most common cause (72.1%) of asymptomatic non-transient proteinuria [4]. Since OP is a benign disease with a relatively good prognosis and often resolves in adulthood, only observation is recommended, without any special treatment. Although the exact etiology is unknown, it is known to be caused by a derangement in the hemodynamic mechanisms and may be accompanied by the nutcracker phenomenon. OP can be classified into transient and stationary reproducible OP, which can lead to persistent proteinuria and cause glomerulopathy [3,6-9]. This report describes a pediatric case of asymptomatic orthostatic proteinuria that progressed to persistent proteinuria with mesangial proliferative glomerular lesions.
Case report
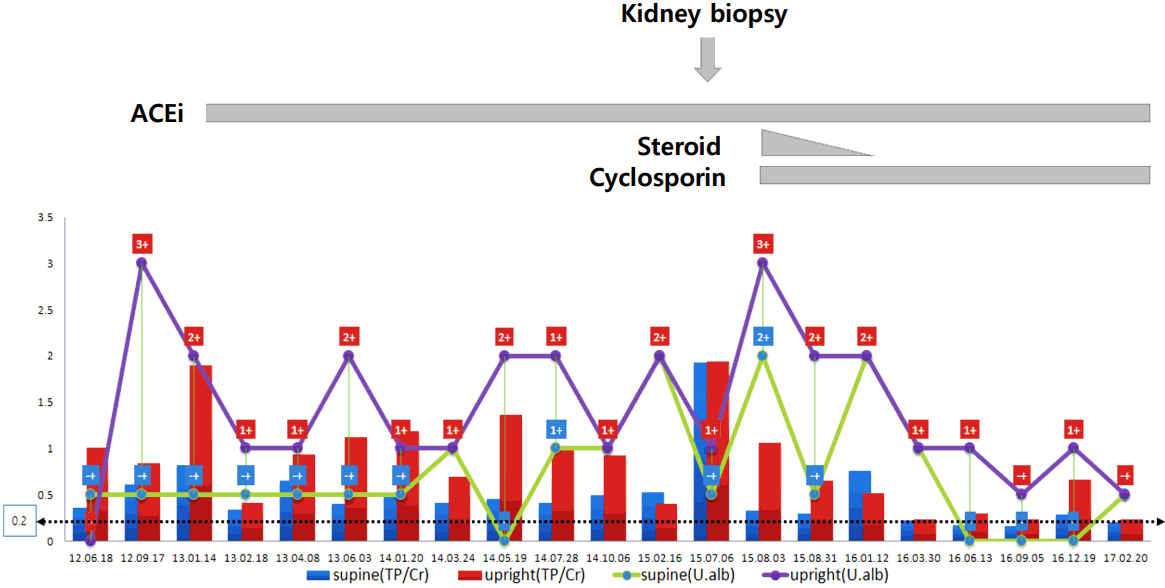
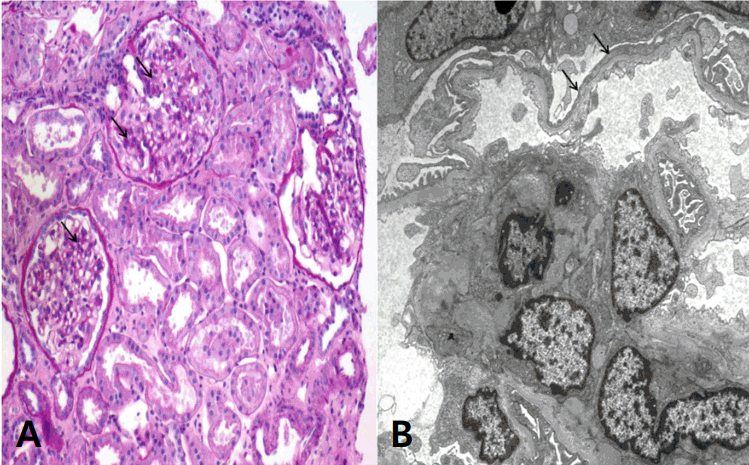
An 11-year-old girl was referred to our hospital for further examination because of proteinuria 2+ found during routine school mass screening urinalysis 1 month earlier. She was well-developed and nourished, with no edema, gross hematuria, joint pain, recent notable infections, rashes, or non-steroidal anti-inflammatory drug use. Her blood pressure was 90/60 mmHg, and her physical examination was unremarkable. Up to now, she has not complained of any abnormal physical symptom or sign. Her serum chemical and immunologic profiles were all within normal limits, and renal ultrasound showed normal kidneys without the nutcracker phenomenon. Initial urinalysis showed specific gravity 1.026 in supine position at early morning urine and 1.013 after 2-hour upright position. And urinalysis was normal pH with equivocal protein (±) in supine position and negative protein in upright position. However, her protein/creatinine ratio (TP/Cr) in supine position and upright position were 0.36 and 1.01 mg/mg, respectively. Eight months later, the protein levels at supine and upright positions changed to ± and 2+, respectively, and the TP/Cr ratio was 0.82 and 1.9 mg/mg in supine and upright positions, respectively. In urine protein electrophoresis, albumin was a predominant fraction (61.3%). Angiotensin II inhibitor treatment was started to reduce proteinuria. During 2 years of observation, her proteinuria was initially thought to orthostatic proteinuria; however, the TP/Cr ratio was 0.53 mg/mg in supine position and 0.4 mg/mg in upright position showed more proteinuria in the supine position. In the urinalysis, proteinuria showed 2+ in both supine and upright positions. Angiotensin II inhibition might not have reduced her proteinuria. The maximal 24-hour urine TP/Cr ratio was 1.9, and the maximal 24-hour urine TP was 830 mg (Fig. 1). Laboratory tests showed normal kidney function, with blood urea nitrogen level of 10.6 mg/dL and Creatinine level of 0.4 mg/dL. Although OP shows favorable prognosis, renal biopsy was performed because of persistent non-orthostatic proteinuria. Light microscopy showed moderate mesangial hypercellularity with slightly increased mesangial matrix, focal tubulointerstitial fibrosis, and atrophy without glomerulosclerosis or crescent formation. Focally marked podocyte foot process effacement was noted, and the glomerular basement membrane had normal thickness with partly irregular inner contours (Fig. 2A, B). Immunofluorescence study showed negative staining. For mesangial proliferative glomerular lesions and persistent non-orthostatic proteinuria, the patient was given low-dose steroid (0.42 mg/kg/day) and cyclosporine (1.5 mg/kg/day) over 6 months. Currently, she is under low-dose cyclosporine and angiotensin II inhibition treatment, and her 24-hour proteinuria shows improvement, with a TP/Cr ratio <0.3 mg/mg (Fig. 1).
Discussion
OP is a non-pathological benign condition of isolated asymptomatic proteinuria. Since OP is a benign disease with a relatively good prognosis and often resolves in adulthood, only observation is recommended, without special treatment [1,2]. OP can be classified into transient and stationary reproducible forms, which can lead persistent proteinuria, and if the level of proteinuria is very high, it may cause glomerulopathy [3,6-9].
Although the exact etiology is unknown, it is known to be caused by hemodynamic mechanisms and may be accompanied with the nutcracker phenomenon. The secretion of angiotensin II and noradrenaline increases in the standing position, which affects the vascular contraction and resistance of efferent arterioles, thereby leading to proteinuria by increasing the glomerular filtration pressure and rate [2,5]. Although the nutcracker phenomenon was not found in this case, it was noted in a considerable number of OP patients, which denoted that hemodynamic mechanisms might be involved in OP development [9,10]. Proteinuria is also caused by or induces underlying subclinical immune injury [11]. As hemodynamic mechanisms, including angiotensin II and proteinuria, per se are important risk factors of renal progression [12-14], prolonged and significant proteinuria can cause glomerular and tubular changes, even though it is orthostatic. Proteinuria leads to transit of protein through glomerular structures, including the glomerular basement membrane, the mesangium, and the podocytes, and increases traffic of protein through the proximal tubules by pinocytosis of filtered protein. This traffic of protein may injure the cells concerned, leading to glomerular and tubular injuries, such as foot process effacement, albumin deposition in podocytes, mesangial changes, and tubulointerstitial inflammation and fibrosis. Therefore, renal pathology in OP can be determined by proteinuria, a protein-overloaded condition, or an underlying subclinical glomerular injury [12-14].
Robinson et al. [6] reported that evidence of a well-defined form of parenchymal renal disease was found in only 8% of 56 patients with fixed and reproducible OP, and 45% revealed distinctive glomerular alterations characterized by capillary wall thickening without basement membrane thickening, focal mesangial hypercellularity, slight capsular thickening, and the presence of eosinophilic granules within the capsular space. Thirty years later, von Bonsdorff et al. [7] reported that they found either normal or slight mesangial proliferation in light microscopy; mesangial, capillary or arteriolar deposits of complement C3 and/or immunoglobulins in 10 of 12 OP cases in immunofluorescence study; and slight subepithelial, intramembranous, and mesangial alterations in electron microscopy; however, focal loss of the podocyte foot processes was seen in only 1 of 15 OP cases. Therefore, they concluded that there was no histological, electron microscopic, or immunohistochemical alterations specific for OP. A rare case of OP presenting with focal segmental glomerulosclerosis with nephrotic syndrome and progressive renal insufficiency developed in a 15-year-old boy who presented initially 1 year earlier with OP [8]. A case of OP associated with nutcracker syndrome showing mesangial hypercellularity was also reported [9]. In this report, a pediatric case of asymptomatic orthostatic proteinuria that progressed to persistent proteinuria and developed mesangial hypercellularity, focal podocyte foot processes effacement, and tubulointerstitial fibrosis and atrophy. The renal pathologic lesions found in this patient may have been induced by persistent moderate to severe proteinuria due to orthostatic proteinuria or by an original glomerular pathology that presented as OP. In conclusion, children with persistent moderate to severe proteinuria should be observed closely because they can’t exclude renal pathology although showing an orthostatic pattern.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.