| Child Kidney Dis > Volume 23(2); 2019 > Article |
|
Abstract
Background
Methods
Results
Conclusion
Notes
Acknowledgement
References
Fig.┬Ā1.
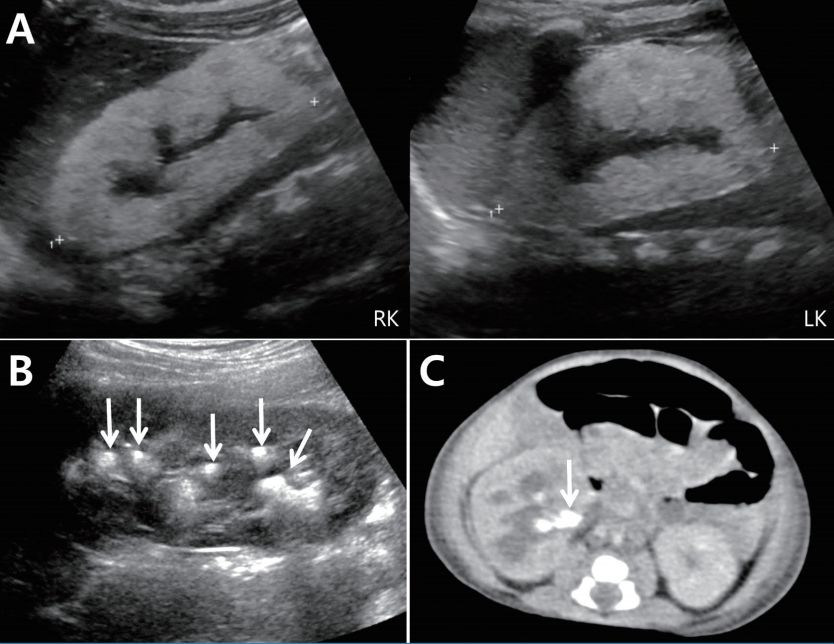
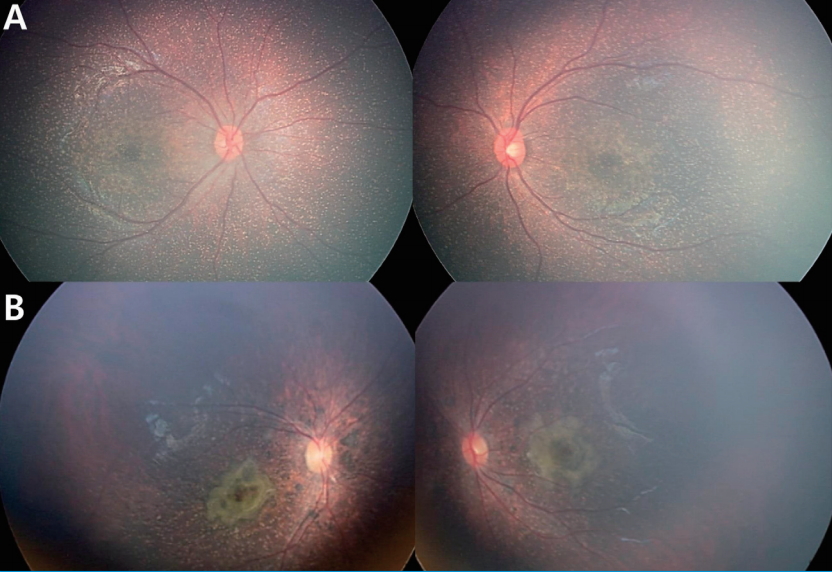
Fig.┬Ā2.
Table┬Ā1.
| Patients | Genes | Mutation 1 | Mutation 2 |
|---|---|---|---|
| 1 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | c.577dupC, p.Leu(CTT)193Pro(CCT)fs |
| 2 | AGXT | c.33delC, p.Lys(AAG)12Arg(AGG)fs*34 | c.335C>A, p.Ala(GCC)112Asp(GAC) |
| 3 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | Homozygote |
| 4 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | c.681-1G>A in intron 6a |
| 5 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | Homozygote |
| 6 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | c.346G>C, p.Gly(GGG)116Arg(CGG) |
| 7 | AGXT | c.331C>T, p.Arg(CGA)111*(TGA) | Homozygote |
| 8 | AGXT | c.187G>C, p.Gly(GGC)63Arg(CGC) | c.823_824dupAG, p.Ser(AGC)275Arg(AGA)fs |
| 9 | AGXT | c.33dupC, p.Lys(AAG)12Gln(CAA)fs | c.568G>A, p.Gly(GGG)190Arg(AGG) |
| 10 | GRHPR | c.181G>A, p.Asp(GAC)61Asn(AAC)* | not detected |
| 11 | HOGA1 | c.812G>A, p.Arg(CGC)271His(CAC)* | not detected |
Table┬Ā2.
GA, gestational age; Dx, diagnosis; eGFR, estimated glomerular filtration rate calculated using the Schwartz formula (ml/min/1.73 m2); ESRD, end-stage renal disease; F-U, follow-up; GHU, gross hematuria; HD, hemodialysis; PD, peritoneal dialysis; RRT, renal replacement therapy; KT, kidney transplantation; LT, liver transplantation; NL, nephrolithiasis; NC, nephrocalcinosis; n.c., not checked; Ox/Cr, oxalate to creatinine ratio (mmol/mol) in spot urine; Scr, serum creatinine (mg/dL).
- TOOLS
-
 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link XML Download
XML Download Full text via DOI
Full text via DOI-
 Download Citation
Download Citation
- Download Citation
-
- Close
 Print
Print-
Share :
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 7,508 View
- 206 Download
- ORCID iDs
-
Yunsoo Choe

https://orcid.org/0000-0001-9396-1676Jiwon M. Lee

https://orcid.org/0000-0003-3932-614XJi Hyun Kim

https://orcid.org/0000-0001-8477-0157Myung Hyun Cho

https://orcid.org/0000-0002-3237-3173Seong Heon Kim

https://orcid.org/0000-0001-8003-3010Joo Hoon Lee

https://orcid.org/0000-0001-8010-3605Young Seo Park

https://orcid.org/0000-0001-7653-2036Hee Gyung Kang

https://orcid.org/0000-0001-8323-5320Il Soo Ha

https://orcid.org/0000-0001-5428-6209Hae Il Cheong

https://orcid.org/0000-0001-7556-1265 - Related articles
-
Parvovirus B19 Infection in Two Korean Pediatric Kidney Transplant Patients2005 October;9(2)
An Experience of Therapeutic Plasma Exchange in 9 Pediatric Patients2005 April;9(1)




