Measurement of urinary protein in children
Article information
Abstract
Proteinuria is an early hallmark of kidney disease and a major risk factor for systemic cardiovascular diseases. There are several methods to measure proteinuria, such as the urine dipstick test, 24-hour urinary protein excretion method, and spot urine for the protein-to-creatinine ratio. The urine dipstick test is simple but inaccurate. The 24-hour urinary protein excretion method is the gold standard; however, it is cumbersome, especially in children. Spot urine for the protein-to-creatinine ratio is simple and accurate, but has limitations. Specific urinary protein such as albumin can be measured instead of the total protein content. Tests should be avoided in situations that cause transient proteinuria or false-positive results. It should be performed correctly, and its limitations should be recognized and interpreted accurately.
Introduction
Proteinuria is a marker of kidney disease that can be detected at an early stage. Additionally, it is an independent risk factor for a decline in kidney function, cardiovascular morbidity, and mortality [1]. Determining the degree of proteinuria is important for early diagnosis and management of kidney disease. Several methods are available for measuring urinary protein levels. There is a difference in how urine and urinary protein are collected and measured. We must understand their meaning, advantages, and disadvantages to anticipate the status and pathophysiology of pediatric patients with proteinuria and proceed with further evaluation. This review discusses the advantages and limitations of several urinary protein measurement methods.
Urine dipstick test
The urine dipstick test is the most convenient method to assess proteinuria. It is inexpensive and can be easily performed using a single urine sample. The procedure can be quickly performed without any apparatus. The dipstick test has a high negative predictive value [2]. It is recommended to screen for proteinuria in pediatric low-risk groups [3].
The urine dipstick test involves colorimetric chemical reactions. The principle is that the presence of negatively charged proteins in a solution alters the color of acid-base indicators. The dipstick is a strip impregnated with buffer and the indicator, tetrabromophenol [4]. The pH of the urine remains constant by means of a buffer, so that the presence of protein is indicated by any change in the color of the indicator. When the dipstick is dipped in urine, the strip can change its color from yellow through green to blue according to the concentration of protein in the urine. Depending on the color, it is reported as negative, trace (10–29 mg/dL), 1+ (30–100 mg/dL), 2+ (100–300 mg/dL), 3+ (300–1,000 mg/dL), or 4+ (>1,000 mg/dL), to give an approximate urinary protein concentration. Reading the results manually rather than using an automated urine dipstick analyzer can lead to overestimation or underestimation.
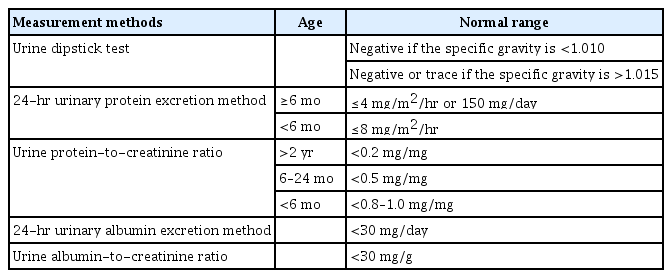
The urine dipstick method is semiquantitative because it does not consider the kidney filtration rate. It is graded based on the urinary protein concentration and cannot represent the amount of urinary protein per day, which is the true diagnostic criterion for proteinuria. Proteinuria has occasionally been defined as ≥1+ [5]. This is equivalent to 300–1,000 mg/m2/day of urinary protein, assuming an average daily urine volume of 1 L/m2. This is higher than the diagnostic criterion for proteinuria (100 mg/m2/day). However, the urine concentration expressed by urine specific gravity should be taken into consideration [6]. A urine dipstick test for protein is considered positive if there is ≥trace in a urine sample in which the specific gravity is <1.010. A dipstick must be ≥1+ to be considered clinically significant if the specific gravity is >1.015 [7]. Furthermore, the reagent on the dipstick is sensitive to albumin but may not detect low concentrations of other types of proteinuria, such as tubular proteinuria and Bence Jones proteins in the urine [8]. It is referred to by its brand name, Albustix. Considering these limitations, there are several conditions under which the dipstick results should be interpreted carefully. False-positive results can occur with highly concentrated urine, a very high urine pH (>7.0), contamination with blood, pus, semen, or vaginal secretions in adolescents, pyuria, or prolonged dipstick immersion [9]. False-negative test results can occur in patients with dilute urine, low urine pH (<4.5), or in disease states in which albumin is not the predominant urinary protein [7].
24-Hour urinary protein excretion method
The gold standard method for the assessment of proteinuria is to measure the amount of protein in a 24-hour urine collection [10]. This is consistent with the diagnostic criteria unit of proteinuria, the amount of urinary protein per day. Variations were quantified using the body surface area. In children over 6 months of age, proteinuria is defined as >4 mg/m2/hr or 150 mg/day [7]. In children under 6 months of age, it is defined as >8 mg/m2/hr [11]. Protein excretion >40 mg/m2/hr is indicative of nephrotic-range proteinuria [11].
However, this is inconvenient, time-consuming, and cumbersome. This is especially difficult for young children who are incontinent and may be unfeasible. Its accuracy depends on the correct collection. Patients were required to empty the bladder and collect all urine for the next 24 hours. At the end of this period, they empty the bladders and save them. It can start at any time; however, it is common to start collection immediately after waking up because the bladder is emptied at a certain time. All the urine samples were stored in a refrigerator. Poor storage results in the growth of alkalizing bacteria in urine samples, which can interfere with the protein evaluation method [11]. To determine whether the specimen was collected for 24 hours properly, the amount of urine creatinine was measured simultaneously. The amount of urine creatinine was estimated as follows: males, 20–25 mg/kg/day; females, 15–20 mg/kg/day [7].
The spot urine protein-to-creatinine ratio (UPCR) described below has largely replaced the 24-hour urinary protein excretion method. However, the 24-hour urinary protein excretion method is still recommended for assessing proteinuria in doubtful situations. It averages any changes in proteinuria according to the circadian rhythm, physical activity, and posture [12].
Urine protein-to-creatinine ratio
UPCR is a fast and simple method for quantitative evaluation of urinary protein excretion. It is useful to screen for children at high risk of proteinuria and to monitor urinary protein levels over time [3]. It is obtained by dividing the urinary protein concentration by the urine creatinine concentration in a spot urine sample. There is no need to consider body surface area in the diagnostic criteria for proteinuria in children because urine creatinine is related to body size [13]. A ratio of <0.2 (mg/mg), the same criteria as adults, is referred to as normal urinary protein excretion in children >2 years of age. Children under the age of 2 years are more likely to lose urinary protein owing to reduced proximal tubule reabsorption [11]. In children 6 to 24 months of age, a ratio of <0.5 is referred to as normal [7]. For infants less than 6 months of age, the normal UPCR is not clearly defined, although a ratio ≥0.8–1.0 is generally considered abnormal [14]. A UPCR of >2 suggests nephrotic-range proteinuria.
Excellent reliability, as well as convenience, makes UPCR an alternative to the 24-hour urinary protein excretion method, which is the gold standard method. It is closely correlated with the amount of protein in 24-hour urine collection in children [15,16]. The log regression analysis of UPCR and the amount of protein in 24-hour urine collection is highly significant from the normal range to nephrotic-range proteinuria [15,16]. Huang et al. [16] showed that a UPCR 0.18 mg/mg, which is close to the cutoff value of 0.2, had high sensitivity and specificity relative to the amount of protein in 24-hour urine collection at 100 mg/m2/day.
However, the UPCR has several limitations. UPCR may not always reflect the 24-hour urinary protein excretion method because protein excretion changes with the time of the day, degree of exercise, and stress level [5]. In addition, it does not consider the deviation of inter-individual differences in daily creatinine excretion. The muscle mass of an individual significantly affects test results. The amount of proteinuria is underestimated in children with high urinary creatinine concentrations, such as athletes. In contrast, it may be overestimated for children with low urinary creatinine concentrations, such as those with muscular dystrophy or severe malnutrition. The degree of dilution also affects UPCR results [17]. It is more likely to overestimate the actual urinary protein excretion in a low-urine-specific gravity sample. Conversely, the UPCR of concentrated urine is likely to be underestimated [17]. The results should be interpreted with caution in newborns and children with polyuria whose urine is not concentrated. If creatinine excretion is not constant, such as in acute kidney injury, UPCR is not useful for estimating the 24-hour excretion rate [18].
Measurement of urinary albumin
Albuminuria is urinary leakage of the specific serum protein, albumin. Only a small fraction of albumin can get filtered in a healthy glomerulus, and most filtered albumin is reabsorbed by the proximal tubules [19]. The compromised integrity of the glomerular filtration barrier is an important etiology of albuminuria [20]. It is independently associated with a higher risk of chronic kidney disease (CKD) progression, cardiovascular events, and death in adults, including diabetes [21]. Many international guidelines now recommend measuring albumin rather than total protein in adults with CKD [22,23].
The measurement of urinary albumin levels has many similarities with urinary protein measurement. Quantification of urinary albumin can be performed using either timed or spot urine collection. The 24-hour urine collection is considered the gold standard method for urinary albumin measurement [24]; however, it is cumbersome, especially in children. Urine albumin-to-creatinine ratio (ACR) is a good alternative. The ACR is consistent with the 24-hour timed urine albumin collection in the pediatric population [25]. Generally, the normal range of albuminuria in children is <30 mg of albumin in the urine per day, or ACR of <30 mg/g in a first-morning urine collection. Moderately increased albuminuria (urinary albumin excretion rate of 30–300 mg/day in 24-hour urine collection, or ACR of 30–300 mg/g in a first-morning urine collection) is usually termed microalbuminuria [26]. Microalbuminuria is defined as an albumin level above the normal level in urine; however, it is below the detectable range in conventional urine dipstick methods [7]. Severely increased albuminuria (urinary albumin excretion rate of >300 mg in a 24-hour urine collection, or ACR >300 mg/g in a first-morning urine collection) is usually termed macroalbuminuria.
In children, proteinuria, rather than albuminuria, is commonly used as a screening tool. However, albuminuria has also been used in some diseases. These include type I diabetes, some parts of Alport syndrome, and autosomal dominant polycystic kidney disease. Pediatric type I diabetes guidelines recommend assessment of albuminuria for CKD screening [27]. In children with Alport syndrome, the onset of microalbuminuria is recommended as a starting treatment for females with X-linked Alport syndrome and for males and females with autosomal dominant Alport syndrome [28]. Children with autosomal dominant polycystic kidney disease and those at risk should be monitored for albuminuria [29]. Some studies have shown that albuminuria in childhood is associated with CKD [30,31]. The 2021 National Institute for Health and Care Excellence guidelines recommend the use of ACR rather than UPCR in children with CKD because ACR has greater sensitivity for low levels of proteinuria than UPCR [22]. Albuminuria measurement is likely to be used more frequently in children.
The timing of the urine sample collection
Most proteinuria in children is transient [7]. Several factors are responsible for transient proteinuria. These include fever, exercise, dehydration, seizures, cold exposure, and/or stress [7]. The amount of urinary protein cannot be measured accurately even when gross hematuria, menstrual contents, or sperm are present in the urine. When these factors are present, physicians should postpone the measurement of urinary protein. If not, we focus on the interpretation of the results. Orthostatic proteinuria is the most common cause of persistent proteinuria in school-aged children and adolescents [7]. Orthostatic proteinuria can occur in preschool children [32]. In children with persistent proteinuria, samples must be collected from their first-morning urine. Children should completely empty their bladder before going to bed and collect the first urine sample as soon as they wake up in the morning. If the results are ambiguous, the examination should be repeated.
Conclusion
Several methods are available for the assessment of proteinuria. They have different normal levels depending on age in children (Table 1). Each method has its strengths and limitations regarding accuracy and convenience (Table 2). These methods should be correctly performed, their limitations should be recognized, and the findings should be interpreted accurately.
Notes
Conflicts of interest
Myung Hyun Cho is an editorial board member of the journal but was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflict of interest relevant to this article was reported.
Funding
None.
Author contributions
All the work was done by MHC.